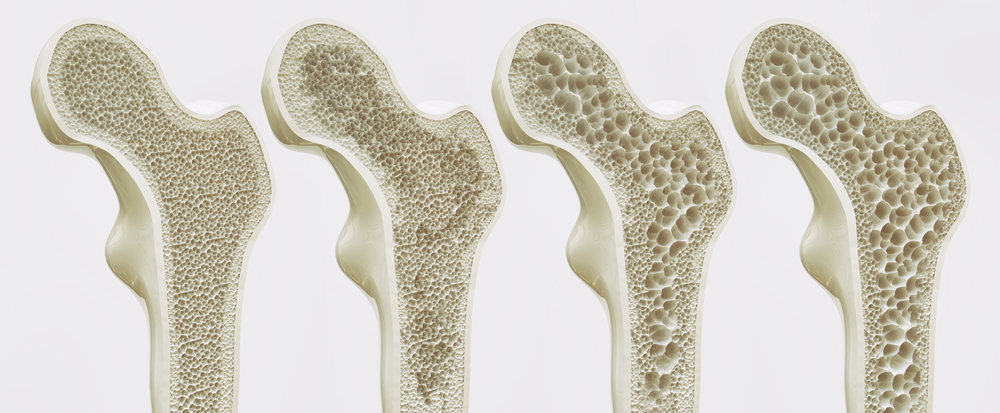
Osteoporosis is the most common age-related bone disease, affecting hundreds of millions of people worldwide. It is estimated that one in three women and one in five men over the age of 50 suffer from osteoporotic fractures. Some risk factors for osteoporosis, such as old age and being female or having a family history of the disease are inevitable. However, negative lifestyle factors such as smoking, drinking alcohol, taking certain medications or exposure to environmental pollutants can be avoided.
Osteoporosis is caused by an excessive activity of bone-resorbing cells while the activity of bone-forming cells is reduced. In healthy people, a tightly controlled process balances the activity of osteoblasts, which build bone, and osteoclasts, which break down bone. However, damage to the cells’ mitochondria can cause this process to go awry, according to new research. The results shed light on how exposure to cigarette smoke, alcohol, and certain medications and environmental toxins can increase the risk of osteoporosis.
Mitochondria Have Impact on Bone Health
The study, led by researchers at Penn’s School of Veterinary Medicine, unveils a mechanism by which these factors and osteoporosis may be linked. Damage to the mitochondria – important cell organelles and energy generators – leads to an increase in the formation of cells called osteoclasts, which are responsible for bone breakdown. These effects could be revealed both in cell cultures and in animal models. Impaired mitochondrial function not only has negative impact on energy production, it also triggers a kind of stress signal that induces overproduction of osteoclasts.
To understand how mitochondrial damage from the work of macrophages might be linked to osteoporosis, the researchers induced damage to a key enzyme responsible for energy production in mitochondria, cytochrome oxidase C, in lab-grown mouse macrophages. This caused the macrophages to release a variety of signaling molecules associated with an inflammatory response and also seemed to encourage them to head towards the osteoclasts.
Looking closely at what was going on, they observed an abnormality with a key molecule, RANK-L, which helps to regulate the bone-building process and is released by bone-building cells to induce bone breakdown. When mitochondria were damaged, they succumbed to stress signals and converted into osteoclasts much faster, even when RANK-L levels were low. These osteoclasts resulted in higher rates of bone resorption or bone loss.
The researchers confirmed their findings in a mouse model, showing that animals with a mutation that leads to dysfunctional mitochondria had increased production of osteoclasts. In addition, their macrophages had higher levels of phagocytosis, the process by which the immune cells engulf and “eat” invaders. The researchers believe this enhanced phagocytic ability may be responsible for some of the other physiological problems that people with defects in their mitochondria can experience.
Because some of the same environmental risk factors that appear to promote osteoporosis, such as smoking and some drugs, can also affect mitochondrial function, the researchers believe these stress signals may be the way through which they affect bone health. Future studies could investigate how maintaining mitochondrial function protects against osteoporosis.
Cx43 Hemi Channels Essential for Bone Strengthening
Physical activities that put mechanical stress on the bones stimulate new bone growth. Osteocytes, which make up 90-95% of all bone cells, help detect mechanical stress on bone. They then regulate the breakdown of weakened bone by cells called osteoclasts and the building of new bone by cells called osteoblasts. Previous research has shown that mechanical stress on bones increases the number of channels on the surface of osteocytes, known as connexin (Cx) 43 hemichannels. In a new study, scientists wanted to find out what role these channels play in the reaction to mechanical stress on the bones.
To do this, they examined two types of mouse models. In one case, the connections between bone cells, called gap junctions, were compromised, but the Cx43 hemichannels were strengthened. In the other, both the gap junctions and the Cx43 half-channels were affected. The team then tested what happens to the bones of these mice, as well as typical mice, when subjected to mechanical stress. They found that a chemical called prostaglandin (PGE2) was produced in both the typical and gap-junction-impaired mice in response to mechanical stress, and their bones became stronger. On the other hand, this response was absent in the Cx43 hemichannel-impaired mice.
To confirm that Cx43 hemichannels are essential for bone strengthening, the team then gave typical mice an antibody that blocks Cx43 hemichannel activity. They found that this also prevented the animals from releasing PGE2 and reduced bone strength in response to mechanical stress. But treating the same mice with PGE2 restored the bone-strengthening effects of mechanical stress.
Existing treatments for osteoporosis generally target osteoclasts, which break down weakened bones, and work by reducing bone turnover. This can unintentionally lead to more brittle bones over time and has been linked to some rare but serious side effects. This research indicates that targeting Cx43 hemichannels to osteocytes could be an alternative treatment option that leverages the body’s response to mechanical stress. This discovery could help scientists develop new treatments for osteoporosis, or other diseases that contribute to bone loss in patients.
New Treatment Strategies for Osteoporosis
Most current osteoporosis therapies involve the use of bisphosphonates, which block the activity of bone resorbing cells and thus prevent excessive bone resorption. However, prolonged treatment with these drugs eliminates the necessary bone remodeling, leading to an increased risk of fractures and other undesirable side effects. Therefore, there is an urgent need to develop new strategies that overcome the limitations of current treatments. There are new advances in this area.
Using genetic analyzes in a small laboratory fish model, the Japanese medaka (Oryzias latipes), researchers identified a small protein, the chemokine CXCL9, that diffuses towards reservoirs containing bone-resorbing cell precursors under osteoporotic conditions. These progenitors produce a receptor, CXCR3, on their cell surface. Upon activation by CXCL9, the precursors are mobilized and migrate in a highly directed manner over long distances toward the bone matrix, where they begin bone resorption.
Both CXCL9 and its receptor CXCR3 have long been known to modulate the migration of immune cells to sites of inflammation, such as in psoriasis and rheumatoid arthritis. There are several chemical inhibitors that block CXCR3 activity that have had little success in clinical trials to treat psoriasis. The research team showed that these inhibitors are highly effective in blocking bone resorbing cell recruitment and protecting bones from osteoporotic damage.
The new strategy allows for fine-tuned modulation of the number of osteoclasts recruited to the bone matrix, rather than widespread blockade of osteoclast activity as with conventional therapies. This has great advantages as excessive bone resorption can be specifically prevented while still maintaining normal bone turnover. This offers potential to avoid increased fracture risks in osteoporosis patients and maintain healthy bone for an improved quality of life.